Health
FDA Grants Accelerated Approval for Jazz Pharma’s Modeyso Therapy
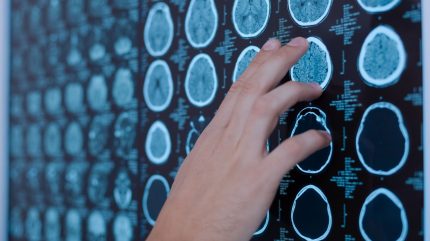
The US Food and Drug Administration (FDA) has granted accelerated approval to Jazz Pharmaceuticals for its innovative treatment, Modeyso (dordaviprone), aimed at addressing diffuse midline gliomas in patients aged one year and older who harbor an H3 K27M mutation. This decision marks a significant milestone as it is the first FDA-approved therapy for this rare and aggressive form of brain tumour.
Modeyso is anticipated to be commercially available by late summer of 2025. Continued approval will depend on the verification of its clinical benefits through the ongoing Phase 3 ACTION confirmatory trial. This trial is assessing both the safety and efficacy of Modeyso in newly diagnosed patients with H3 K27M-mutant diffuse glioma following radiotherapy.
Mechanism and Clinical Insights
Modeyso functions as a protease activator of the mitochondrial caseinolytic protease P (ClpP), inhibiting the dopamine D2 receptor (DRD2). In laboratory studies, it has been shown to activate the integrated stress response, induce apoptosis, and modify mitochondrial metabolism. Notably, it restores histone H3 K27 trimethylation in H3 K27M-mutant diffuse glioma, which is critical for managing tumour growth.
The FDA’s decision was based on an integrated efficacy analysis involving 50 patients with recurrent H3 K27M-mutant diffuse midline glioma, drawn from five open-label studies. The overall response rate (ORR) reported was 22%, with a median duration of response lasting 10.3 months. Among those who responded to the treatment, 73% maintained their response for at least six months.
Expert Opinions and Safety Profile
Joshua E Allen, Chief Scientific Officer of Chimerix, a subsidiary of Jazz Pharmaceuticals, expressed the significance of this approval, stating, “The FDA approval of Modeyso is a milestone moment for the patients and families who have long needed new options, the clinicians who have tirelessly searched for solutions, and the researchers and advocates who never gave up.” Allen emphasized that this approval not only provides clinicians with the first targeted option for this disease but also signifies a meaningful shift in patient expectations following diagnosis.
The safety profile of Modeyso was evaluated in a broader patient population, involving 376 patients across four open-label clinical studies. Serious adverse events were reported in 33% of these patients. Common side effects included fatigue, headache, vomiting, nausea, and musculoskeletal pain.
Diffuse midline glioma with the H3 K27M mutation is characterized by a specific genetic alteration that disrupts epigenetic regulation and drives tumour growth. This rare and aggressive brain tumour predominantly affects children and young adults, highlighting the urgent need for effective treatment options.
As the landscape of cancer therapies evolves, the approval of Modeyso represents a crucial development in the fight against diffuse midline glioma, providing hope to patients and families affected by this challenging condition.
-

 Health3 months ago
Health3 months agoNeurologist Warns Excessive Use of Supplements Can Harm Brain
-

 Health3 months ago
Health3 months agoFiona Phillips’ Husband Shares Heartfelt Update on Her Alzheimer’s Journey
-

 Science1 month ago
Science1 month agoBrian Cox Addresses Claims of Alien Probe in 3I/ATLAS Discovery
-

 Science1 month ago
Science1 month agoNASA Investigates Unusual Comet 3I/ATLAS; New Findings Emerge
-

 Science4 weeks ago
Science4 weeks agoScientists Examine 3I/ATLAS: Alien Artifact or Cosmic Oddity?
-
 Science4 weeks ago
Science4 weeks agoNASA Investigates Speedy Object 3I/ATLAS, Sparking Speculation
-

 Entertainment4 months ago
Entertainment4 months agoKerry Katona Discusses Future Baby Plans and Brian McFadden’s Wedding
-

 Entertainment4 months ago
Entertainment4 months agoEmmerdale Faces Tension as Dylan and April’s Lives Hang in the Balance
-

 World3 months ago
World3 months agoCole Palmer’s Cryptic Message to Kobbie Mainoo Following Loan Talks
-
 Science4 weeks ago
Science4 weeks agoNASA Scientists Explore Origins of 3I/ATLAS, a Fast-Moving Visitor
-

 Entertainment4 months ago
Entertainment4 months agoLove Island Star Toni Laite’s Mother Expresses Disappointment Over Coupling Decision
-

 Entertainment3 months ago
Entertainment3 months agoMajor Cast Changes at Coronation Street: Exits and Returns in 2025









